 
This model helps in finding the structures of both molecules, having a central metallic atom and a non-metallic central atom. The 3-D shapes and structures of several molecules cannot be determined by Lewis electron-pair theory, and therefore the VSEPR model is used. The arrangement of angles from highest to lowest is determined by the hierarchy of repulsion where lone pair-lone pair is the highest then comes lone pair-bonding which is slightly lower, and bonding-bonding is the least. 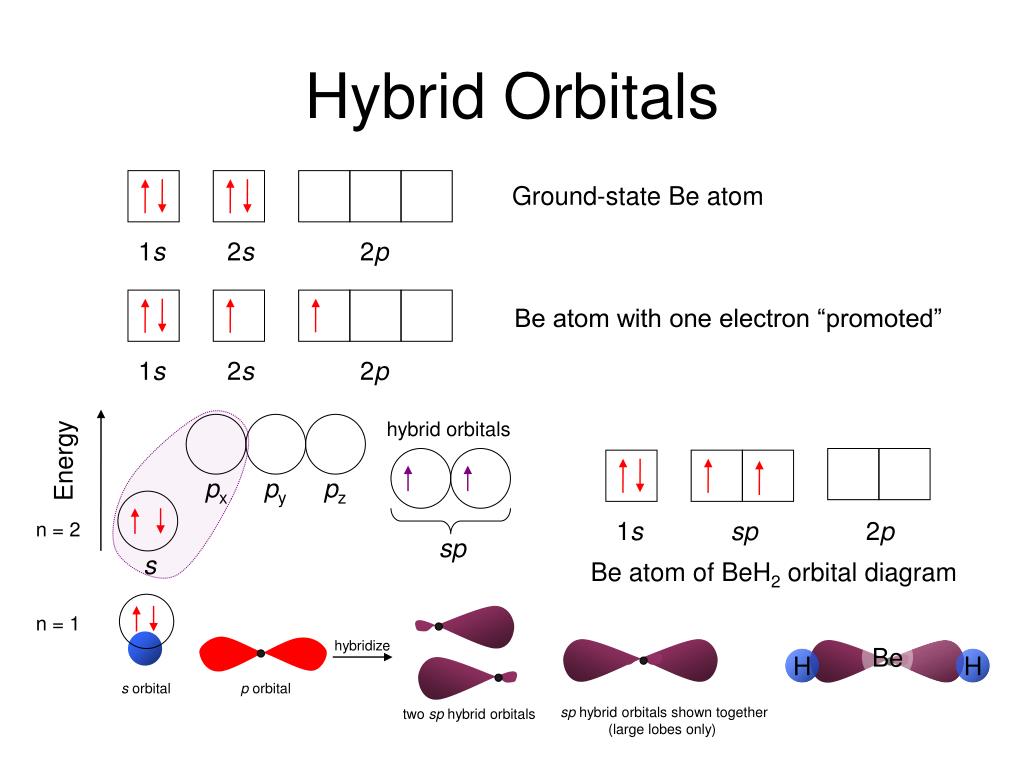
The electronic geometry also determines the angles between the electron domains. For example, a steric number of two gives a linear electronic structure. The sum called the steric number determines the electronic shape of the molecule. Follow the below steps,įind out the sum of the total number of lone pairs and the number of binding domains. Knowing this, the electron geometry can be obtained. Electron domain is nothing but the number of bonds and lone pairs in an atom. Before starting to use the VSEPR model, the Lewis dot picture is considered to determine the electron domain. To reduce the electrostatic repulsion between electron pair is what the theory is based on. The VSEPR model is a straightforward yet useful way to understand and explain the shapes and structure of molecules. But this model does not say anything regarding the multiple bonds present or the bond length. The VSEPR model helps to understand the different shapes and arrangement of molecules. However, this theory provides no information on how the atoms are arranged. To find the types and number of atomic bonds in a substance and also to show which atoms have lone pairs of electrons, the Lewis electron-pair theory can be used. For example, an oxygen molecule is made of two atoms of oxygen, but sometimes under different environmental conditions, oxygen forms a molecule consisting of three atoms of oxygen, called Ozone (O 3 ). They have different sizes and structures. Molecules, as mentioned earlier, are made up of atoms. Co-ordinate bond is a type of covalent bond where the electrons being shared are contributed from one atom only. Quantum theory explains all kinds of bonding, but the simpler explanation is given by the octet rule and VSEPR theory.Įlectron sharing involves the “sharing” of one or more electrons between the atoms involved in the bond formation, whereas electron exchange is the exchange of electrons between atoms and not the sharing. Strong chemical bonding is due to the sharing or transfer of electrons between the atoms involved. The strength of a chemical bond varies from strong or primary bonds and weak or secondary bonds. Chemical bonds can result from three types of interactions between atoms: Sometimes, atoms of particular elements easily bond with different atoms to form molecules. It is based on the assumption that the molecule will take such a shape that will minimize electronic repulsion in the valence shell of that atom.Ītoms come together to form molecules and molecules come together and bind by chemical bonds to form elements or compounds. The theory was given by Sidgwick and Powell in the year 1940. The VSEPR theory is a tool that is used for predicting the shape of a molecule from the electron pairs that surround the central atoms of that molecule. VSEPR theory suggests that the repulsion between two electrons is caused by the Pauli exclusion principle that has greater importance than electrostatic repulsion in the determination of a molecule's geometry. This theory is also known as the Gillespie-Nyholm theory to honor Ronald Nyholm and Ronald Gillespie who laid its foundation. This arrangement of the atom shapes the geometry of the resulting molecule. The VSEPR Theory (Valence Shell Electron Pair Repulsion Theory) is based on the fact that there is a repulsion between the pairs of valence electrons in all the atoms, and the atoms will always try to arrange themselves in a manner in which the electron pair repulsion is minimized. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
